 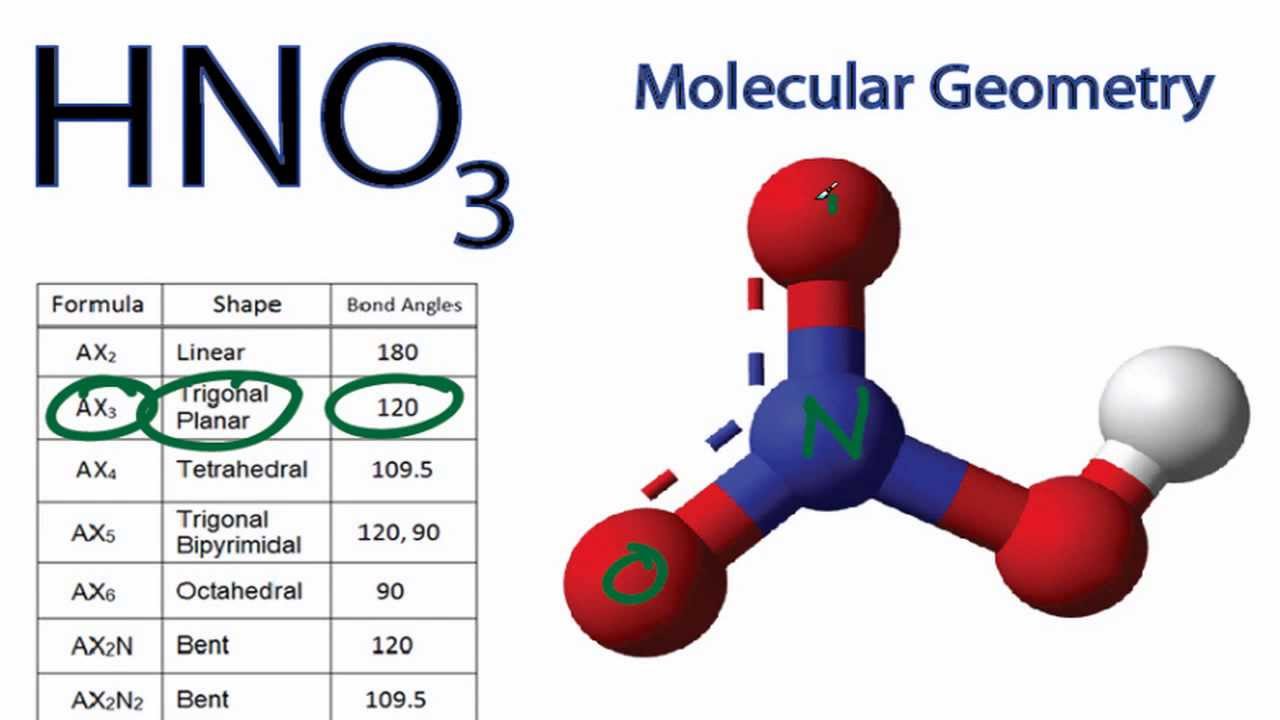 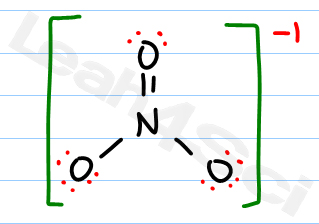
Spectroscopic data, such as infrared and Raman spectroscopy, also supports the hybridization of the central nitrogen atom in NO3. This hybridization results in the formation of three hybrid orbitals with a trigonal planar arrangement, which allows the three oxygen atoms to be positioned around the central nitrogen atom at an angle of 120 degrees. The trigonal planar molecular geometry of NO3 is consistent with the hybridization of the central nitrogen atom as sp2. The evidence for the hybridization of the central nitrogen atom in NO3 comes from various sources, such as the molecular geometry and bond angles of the molecule, as well as spectroscopic data. The third 2p orbital remains unhybridized and contains a lone pair of electrons. To form three hybrid orbitals for bonding with the three oxygen atoms, the 2s and two of the 2p orbitals combine, resulting in three sp2 hybrid orbitals with a trigonal planar arrangement. In NO3, the central nitrogen atom has one 2s and three 2p orbitals available for hybridization. The hybridization of an atom is the mixing of atomic orbitals to form new hybrid orbitals with different energies and shapes that can better explain the geometry of the molecule. To determine the hybridization of the central nitrogen atom in NO3, we can use the concept of hybrid orbitals. In NO3, the central nitrogen atom is surrounded by three oxygen atoms in a trigonal planar arrangement. The hybridization of the NO3 molecule can be determined by analyzing the arrangement of atoms and the lone pairs of electrons around the central nitrogen atom. However, overall, the VSEPR theory provides a good framework for predicting the bond angles of molecules like NO3 based on their molecular geometry. The small deviation between the predicted and observed bond angles of NO3 can be attributed to various factors, such as lone pair effects and hybridization of the central nitrogen atom. This bond angle is in agreement with the observed bond angle of NO3, which has been experimentally determined to be 120.5 degrees. The predicted bond angle for the NO3 molecule, based on its trigonal planar molecular geometry, is 120 degrees. Comparison of predicted and observed bond angles of NO3 This arrangement results in a bond angle of 120 degrees between each oxygen atom and the central nitrogen atom. These bonding pairs repel each other equally, creating a trigonal planar arrangement. In NO3, there are three bonding pairs of electrons and no lone pairs of electrons on the central nitrogen atom. According to this theory, the electron pairs around the central atom repel each other and try to stay as far apart as possible. To determine the shape of the NO3 molecule, we can use the VSEPR (Valence Shell Electron Pair Repulsion) theory. This gives NO3 a trigonal planar molecular geometry. 
In NO3, the central nitrogen atom is surrounded by three oxygen atoms. The molecular geometry of NO3 can be determined by analyzing the arrangement of atoms and lone pairs of electrons around the central nitrogen atom. Determination of the shape of NO3 molecule This polarity makes it capable of forming hydrogen bonds and interacting with other polar molecules. Therefore, the NO3 molecule is polar, as it has a net dipole moment due to the uneven distribution of charge. This results in the oxygen atoms carrying partial negative charges, while the nitrogen carries a partial positive charge. Since the electronegativity of nitrogen is greater than that of oxygen, the electrons in the covalent bonds are more strongly attracted to the nitrogen atom. In NO3, the central nitrogen atom is surrounded by three oxygen atoms, which creates a trigonal planar molecular geometry. The polarity of the NO3 molecule depends on the difference in electronegativity between the atoms and the molecular geometry. Explanation of the polar/non-polar nature of NO3 molecule Check the Lewis structure for formal charges on each atom to confirm its stability.Ĭ.Ensure that the total number of electrons used equals the number determined in step 1.If there are leftover electrons after step 4, add them to the central nitrogen atom to satisfy the octet or sextet rule.Distribute the remaining valence electrons to each atom to satisfy the octet rule, which means each atom (except nitrogen) should have eight electrons in its outermost shell.Connect each oxygen (O) atom to the central nitrogen atom using single bonds.Identify the central atom of the molecule, which in this case is nitrogen (N).Determine the total number of valence electrons in the NO3 molecule by adding up the valence electrons of each atom. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
